Alfons Hoekstra et al. published a new paper in Frontiers in Neurology.
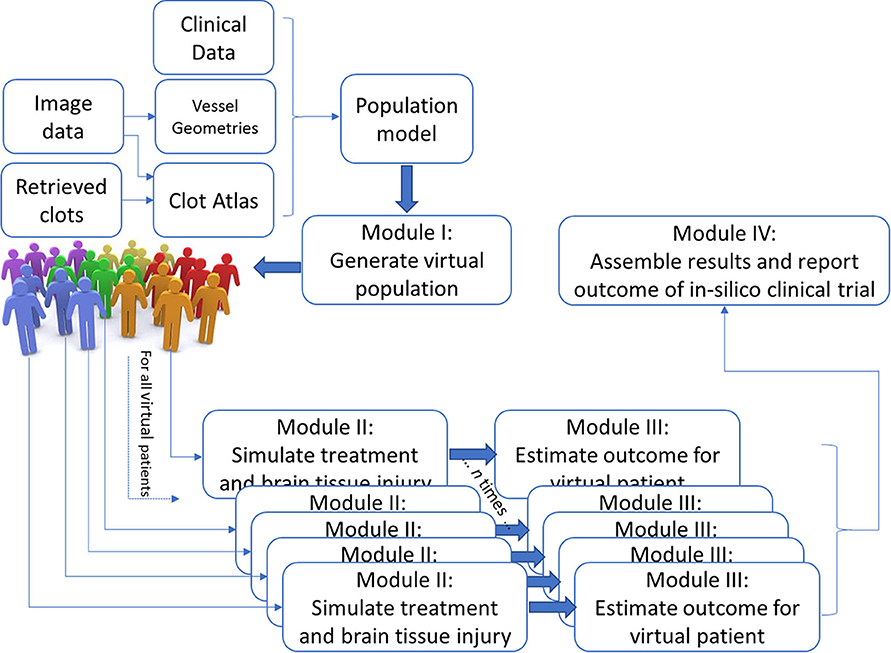
Despite improved treatment, a large portion of patients with acute ischemic stroke due to a large vessel occlusion have poor functional outcome. Further research exploring novel treatments and better patient selection has therefore been initiated. The feasibility of new treatments and optimized patient selection are commonly tested in extensive and expensive randomized clinical trials. in-silico trials, computer-based simulation of randomized clinical trials, have been proposed to aid clinical trials. In this white paper, we present our vision and approach to set up in-silico trials focusing on treatment and selection of patients with an acute ischemic stroke. The INSIST project (IN-Silico trials for treatment of acute Ischemic STroke, www.insist-h2020.eu) is a collaboration of multiple experts in computational science, cardiovascular biology, biophysics, biomedical engineering, epidemiology, radiology, and neurology. INSIST will generate virtual populations of acute ischemic stroke patients based on anonymized data from the recent stroke trials and registry, and build on the existing and emerging in-silico models for acute ischemic stroke, its treatment (thrombolysis and thrombectomy) and the resulting perfusion changes. These models will be used to design a platform for in-silico trials that will be validated with existing data and be used to provide a proof of concept of the potential efficacy of this emerging technology. The platform will be used for preliminary evaluation of the potential suitability and safety of medication, new thrombectomy device configurations and methods to select patient subpopulations for better treatment outcome. This could allow generating, exploring and refining relavant hypotheses on potential causal pathways (which may follow from the evidence obtained from clinical trials) and improving clinical trial design. Importantly, the findings of the in-silico trials will require validation under the controlled settings of randomized clinical trials.
For further details please see: https://www.frontiersin.org/articles/10.3389/fneur.2020.558125/full
